Is It Best to Use Variable Speed for Foam Houses

Cream is an object formed by trapping pockets of gas in a liquid or solid.[1] [2] [3]
A bathroom sponge and the head on a glass of beer are examples of foams. In most foams, the book of gas is large, with thin films of liquid or solid separating the regions of gas. Soap foams are also known every bit suds.
Solid foams can exist closed-jail cell or open-cell. In airtight-cell foam, the gas forms discrete pockets, each completely surrounded by the solid fabric. In open-cell foam, gas pockets connect to each other. A bath sponge is an instance of an open-cell foam: h2o hands flows through the unabridged structure, displacing the air. A camping mat is an instance of a airtight-cell foam: gas pockets are sealed from each other so the mat cannot soak up h2o.
Foams are examples of dispersed media. In general, gas is present, and then it divides into gas bubbles of different sizes (i.eastward., the material is polydisperse)—separated by liquid regions that may class films, thinner and thinner when the liquid phase drains out of the system films.[iv] When the principal scale is minor, i.due east., for a very fine cream, this dispersed medium can be considered a blazon of colloid.
Foam tin also refer to something that is analogous to foam, such as quantum cream, polyurethane cream (foam rubber), XPS foam, polystyrene, phenolic, or many other manufactured foams.
Construction [edit]
A cream is, in many cases, a multi-scale system.

Gild and disorder of bubbles in a surface foam
One scale is the bubble: cloth foams are typically disordered and take a variety of bubble sizes. At larger sizes, the report of idealized foams is closely linked to the mathematical problems of minimal surfaces and three-dimensional tessellations, also chosen honeycombs. The Weaire–Phelan structure is considered the all-time possible (optimal) unit cell of a perfectly ordered cream,[5] while Plateau'due south laws describe how lather-films form structures in foams.
At lower calibration than the bubble is the thickness of the moving-picture show for metastable foams, which can be considered a network of interconnected films called lamellae. Ideally, the lamellae connect in triads and radiate 120° outward from the connection points, known equally Plateau borders.
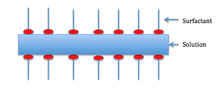
An even lower scale is the liquid–air interface at the surface of the pic. Near of the time this interface is stabilized by a layer of amphiphilic construction, ofttimes made of surfactants, particles (Pickering emulsion), or more complex associations.
Mechanical properties of solid foams [edit]
Solid foams, both open up-cell and closed-jail cell, are considered equally a sub-class of cellular structures. They often have lower nodal connectivity as compared to other cellular structures like honeycombs and truss lattices, and thus, their failure mechanism is dominated by angle of members. Depression nodal connectivity and the resulting failure mechanism ultimately lead to their lower mechanical strength and stiffness compared to honeycombs and truss lattices.[6] [vii]
Formation [edit]
Several conditions are needed to produce foam: there must exist mechanical work, surface active components (surfactants) that reduce the surface tension, and the formation of cream faster than its breakdown. To create cream, work (Westward) is needed to increase the surface area (ΔA):
where γ is the surface tension.
1 of the ways cream is created is through dispersion, where a big corporeality of gas is mixed with a liquid. A more than specific method of dispersion involves injecting a gas through a hole in a solid into a liquid. If this process is completed very slowly, then one bubble can exist emitted from the orifice at a time as shown in the pic below.
One of the theories for determining the separation time is shown beneath; nevertheless, while this theory produces theoretical information that matches with the experimental data, detachment due to capillarity is accepted equally a better caption.

Ascent chimera from orifice
The buoyancy force acts to heighten the bubble, which is
where is the volume of the bubble, is the acceleration due to gravity, and ρ1 is the density of the gas ρtwo is the density of the liquid. The force working against the buoyancy force is the surface tension force, which is
- ,
where γ is the surface tension, and is the radius of the orifice. Equally more than air is pushed into the bubble, the buoyancy forcefulness grows quicker than the surface tension force. Thus, detachment occurs when the buoyancy forcefulness is large enough to overcome the surface tension forcefulness.
In add-on, if the bubble is treated as a sphere with a radius of and the book is substituted in to the equation above, separation occurs at the moment when
Examining this phenomenon from a capillarity viewpoint for a bubble that is being formed very slowly, it can be assumed that the pressure inside is constant everywhere. The hydrostatic pressure in the liquid is designated by . The change in pressure across the interface from gas to liquid is equal to the capillary pressure; hence,
where Rone and R2 are the radii of curvature and are set as positive. At the stem of the chimera, R3 and R4 are the radii of curvature also treated as positive. Hither the hydrostatic pressure in the liquid has to take in account z, the distance from the top to the stem of the bubble. The new hydrostatic pressure level at the stem of the chimera is p 0(ρ i −ρ ii)z. The hydrostatic pressure balances the capillary force per unit area, which is shown beneath:
Finally, the difference in the top and bottom pressure level equal the change in hydrostatic pressure:
At the stem of the bubble, the shape of the bubble is nearly cylindrical; consequently, either R3 or R4 is big while the other radius of curvature is minor. As the stalk of the bubble grows in length, it becomes more unstable equally one of the radius grows and the other shrinks. At a certain point, the vertical length of the stem exceeds the circumference of the stem and due to the buoyancy forces the bubble separates and the procedure repeats.[8]
Stability [edit]
Stabilization [edit]

Marangoni upshot of a film
Marangoni effect of a film (2)
The stabilization of a foam is caused by van der Waals forces between the molecules in the foam, electric double layers created past dipolar surfactants, and the Marangoni effect, which acts as a restoring force to the lamellae.
The Marangoni outcome depends on the liquid that is foaming being impure. Generally, surfactants in the solution decrease the surface tension. The surfactants besides clump together on the surface and class a layer equally shown below.
For the Marangoni effect to occur, the foam must be indented as shown in the first motion picture. This indentation increases local surface area. Surfactants have a larger improvidence fourth dimension than the bulk of the solution—so the surfactants are less full-bodied in the indentation.
Also, surface stretching makes the surface tension of the indented spot greater than the surrounding area. Consequentially—since diffusion time for the surfactants is large—the Marangoni upshot has time to accept place. The difference in surface tension creates a gradient, which instigates fluid flow from areas of lower surface tension to areas of higher surface tension. The 2nd picture shows the film at equilibrium later the Marangoni event has taken identify.[9]
Destabilization [edit]
Witold Rybczynski and Jacques Hadamard developed an equation to calculate the velocity of bubbles that rise in foam with the assumption that the bubbles are spherical with a radius .
with velocity in units of centimeters per second. ρ1 and ρii is the density for a gas and liquid respectively in units of thousand/cm3 and ῃ1 and ῃii is the viscosity of the gas and liquid g/cm·due south and 1000 is the dispatch in units of cm/s2.
However, since the density and viscosity of a liquid is much greater than the gas, the density and viscosity of the gas tin be neglected, which yields the new equation for velocity of bubbles rising equally:
Notwithstanding, through experiments information technology has been shown that a more authentic model for bubbling ascent is:
Deviations are due to the Marangoni consequence and capillary pressure, which impact the assumption that the bubbling are spherical. For laplace pressure of a curved gas liquid interface, the two principal radii of curvature at a point are R1 and Rtwo.[ten] With a curved interface, the pressure in one stage is greater than the pressure in some other phase. The capillary force per unit area Pc is given past the equation of:
- ,
where is the surface tension. The chimera shown below is a gas (phase 1) in a liquid (phase 2) and point A designates the top of the bubble while point B designates the lesser of the bubble.
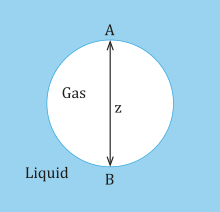
Chimera for hydrostatic force per unit area
At the top of the chimera at indicate A, the pressure in the liquid is assumed to exist p0 equally well as in the gas. At the bottom of the bubble at point B, the hydrostatic pressure is:
where ρ1 and ρtwo is the density for a gas and liquid respectively. The difference in hydrostatic pressure level at the acme of the bubble is 0, while the deviation in hydrostatic force per unit area at the bottom of the bubble beyond the interface is gz(ρ 2 −ρ 1). Assuming that the radii of curvature at point A are equal and denoted by RA and that the radii of curvature at point B are equal and denoted by RB, and so the difference in capillary pressure betwixt point A and indicate B is:
At equilibrium, the difference in capillary pressure must be balanced past the divergence in hydrostatic pressure level. Hence,
Since, the density of the gas is less than the density of the liquid the left hand side of the equation is e'er positive. Therefore, the inverse of RA must be larger than the RB. Meaning that from the pinnacle of the chimera to the lesser of the bubble the radius of curvature increases. Therefore, without neglecting gravity the bubbles cannot be spherical. In addition, as z increases, this causes the departure in RA and RB too, which ways the bubble deviates more than from its shape the larger it grows.[8]
Foam destabilization occurs for several reasons. First, gravitation causes drainage of liquid to the foam base of operations, which Rybczynski and Hadamar include in their theory; however, foam also destabilizes due to osmotic pressure causes drainage from the lamellas to the Plateau borders due to internal concentration differences in the cream, and Laplace force per unit area causes diffusion of gas from small to large bubbles due to pressure deviation. In addition, films can break under disjoining pressure, These effects can pb to rearrangement of the foam structure at scales larger than the bubbles, which may be private (T1 procedure) or collective (even of the "avalanche" blazon).
Experiments and characterizations [edit]
Being a multiscale system involving many phenomena, and a versatile medium, cream tin can be studied using many different techniques. Considering the different scales, experimental techniques are diffraction ones, mainly light scattering techniques (DWS, see below, static and dynamic light scattering, 10 rays and neutron scattering) at sub-micrometer scales, or microscopic ones. Considering the organisation as continuous, its bulk properties can exist characterized past lite transmittance but too conductimetry. The correlation between construction and bulk is evidenced more accurately by acoustics in particular. The organization betwixt bubbles has been studied numerically using sequential attempts of evolution of the minimum surface energy either at random (Pott's model) or deterministic way (surface evolver). The evolution with time (i.east., the dynamics) can exist faux using these models, or the bubble model (Durian), which considers the movement of individual bubbles.
Observations of the pocket-size-scale construction can be made by shining the foam with laser light or x-ray beams and measuring the reflectivity of the films between bubbles. Observations of the global structure can exist done using neutron scattering.

Reflection of radiation by a foam

Measurement principle of multiple light scattering coupled with vertical scanning
A typical light scattering (or diffusion) optical technique, multiple light scattering coupled with vertical scanning, is the about widely used technique to monitor the dispersion state of a product, hence identifying and quantifying destabilization phenomena.[11] [12] [13] [14] It works on any concentrated dispersions without dilution, including foams. When light is sent through the sample, it is backscattered by the bubbles. The backscattering intensity is directly proportional to the size and volume fraction of the dispersed phase. Therefore, local changes in concentration (drainage, syneresis) and global changes in size (ripening, coalescence) are detected and monitored.
Applications [edit]
Liquid foams [edit]
Liquid foams can be used in fire retardant foam, such as those that are used in extinguishing fires, peculiarly oil fires.
In some ways, leavened bread is a foam, every bit the yeast causes the staff of life to ascension by producing tiny bubbles of gas in the dough. The dough has traditionally been understood as a closed-jail cell cream, in which the pores practice not connect with each other. Cutting the dough releases the gas in the bubbles that are cut, but the gas in the balance of the dough cannot escape. When dough is allowed to rise also far, it becomes an open-cell foam, in which the gas pockets are connected. Cutting the dough or the surface otherwise breaking at that bespeak would cause a big book of gas to escape, and the dough would collapse. The open construction of an over-risen dough is piece of cake to detect: instead of consisting of discrete gas bubbles, the dough consists of a gas infinite filled with threads of the flour-water paste. Recent research has indicated that the pore construction in bread is 99% interconnected into 1 large vacuole, thus the closed-cell foam of the moist dough is transformed into an open cell solid foam in the bread.[15]
The unique belongings of gas-liquid foams having very high specific surface area is exploited in the chemical processes of froth flotation and cream fractionation.
Solid foams [edit]
Solid foams are a class of lightweight cellular engineering materials. These foams are typically classified into two types based on their pore structure: open-cell-structured foams (also known every bit reticulated foams) and airtight-prison cell foams. At high enough prison cell resolutions, any type can be treated as continuous or "continuum" materials and are referred to as cellular solids,[16] with predictable mechanical properties.
Open-cell-structured foams comprise pores that are connected to each other and form an interconnected network that is relatively soft. Open-cell foams fill with whatever gas surrounds them. If filled with air, a relatively good insulator results, just, if the open cells fill up with h2o, insulation properties would be reduced. Recent studies have put the focus on studying the properties of open-prison cell foams as an insulator material. Wheat gluten/TEOS bio-foams have been produced, showing similar insulator properties as for those foams obtained from oil-based resources.[17] Foam rubber is a blazon of open-jail cell foam.
Closed-jail cell foams do not have interconnected pores. The closed-jail cell foams usually take higher compressive forcefulness due to their structures. However, airtight-prison cell foams are also, in general more than dense, crave more fabric, and as a consequence are more expensive to produce. The closed cells can exist filled with a specialized gas to provide improved insulation. The closed-cell structure foams have higher dimensional stability, low moisture absorption coefficients, and higher force compared to open-cell-structured foams. All types of foam are widely used every bit core material in sandwich-structured blended materials.
The primeval known engineering use of cellular solids is with wood, which in its dry form is a closed-jail cell cream equanimous of lignin, cellulose, and air. From the early 20th century, diverse types of especially manufactured solid foams came into use. The depression density of these foams makes them excellent as thermal insulators and flotation devices and their lightness and compressibility make them platonic equally packing materials and stuffings.
An instance of the utilize of azodicarbonamide[xviii] as a blowing agent is found in the manufacture of vinyl (PVC) and EVA-PE foams, where it plays a role in the germination of air bubbling by breaking downwardly into gas at loftier temperature.[19] [xx] [21]
The random or "stochastic" geometry of these foams makes them good for energy absorption, besides. In the late 20th century to early 21st century, new manufacturing techniques have allowed for geometry that results in splendid strength and stiffness per weight. These new materials are typically referred to as engineered cellular solids.[sixteen]
Syntactic foam [edit]
A special grade of closed-cell foams, known equally syntactic cream, contains hollow particles embedded in a matrix material. The spheres tin can exist made from several materials, including glass, ceramic, and polymers. The advantage of syntactic foams is that they have a very high forcefulness-to-weight ratio, making them ideal materials for many applications, including deep-sea and space applications. One detail syntactic foam employs shape memory polymer as its matrix, enabling the foam to take on the characteristics of shape memory resins and composite materials; i.due east., it has the ability to be reshaped repeatedly when heated above a certain temperature and cooled. Shape memory foams have many possible applications, such as dynamic structural support, flexible foam core, and expandable foam fill.
Integral skin foam [edit]
Integral skin foam, besides known equally self-skin foam, is a type of foam with a high-density skin and a low-density cadre. It tin be formed in an open-mold procedure or a airtight-mold process. In the open up-mold process, two reactive components are mixed and poured into an open up mold. The mold is and so closed and the mixture is allowed to expand and cure. Examples of items produced using this process include arm rests, baby seats, shoe soles, and mattresses. The closed-mold process, more usually known equally reaction injection molding (RIM), injects the mixed components into a airtight mold nether high pressures.[22]
Defoaming [edit]
Foam, in this instance significant "bubbly liquid", is too produced as an often-unwanted past-production in the industry of various substances. For example, cream is a serious problem in the chemical industry, especially for biochemical processes. Many biological substances, for instance proteins, easily create foam on agitation or aeration. Cream is a problem because it alters the liquid flow and blocks oxygen transfer from air (thereby preventing microbial respiration in aerobic fermentation processes). For this reason, anti-foaming agents, similar silicone oils, are added to prevent these problems. Chemical methods of foam control are not e'er desired with respect to the problems (i.due east., contamination, reduction of mass transfer) they may cause especially in food and pharmaceutical industries, where the production quality is of neat importance. Mechanical methods to forestall foam formation are more than common than chemical ones.
Speed of audio [edit]
The acoustical property of the speed of sound through a foam is of involvement when analyzing failures of hydraulic components. The analysis involves calculating full hydraulic cycles to fatigue failure. The speed of sound in a foam is determined by the mechanical properties of the gas creating the foam: oxygen, nitrogen, or combinations.
Bold that the speed of audio is based on the liquid's fluid backdrop leads to errors in calculating fatigue cycles and failure of mechanical hydraulic components. Using acoustical transducers and related instrumentation that set depression limits (0–50,000 Hz with curlicue-off) causes errors. The depression roll-off during measurement of bodily frequency of acoustic cycles results in miscalculation due to actual hydraulic cycles in the possible ranges of 1–1000 MHz or college. Instrumentation systems are most revealing when wheel bandwidths exceed the actual measured cycles by a factor of x to 100. Associated instrumentation costs as well increase by factors of ten to 100.
Nearly moving hydro-mechanical components cycle at 0–50 Hz, only entrained gas bubbles resulting in a foamy condition of the associated hydraulic fluid results in bodily hydraulic cycles that can exceed 1000 MHz fifty-fifty if the moving mechanical components exercise not cycle at the higher cycle frequency.
Gallery [edit]
Foam scales and properties [edit]
See also [edit]
- Aluminium foam sandwich
- Ballistic foam
- Cellular Solids
- Chaotic bubble
- Metal foam
- Nanofoam
- Sea foam
- Reversibly assembled cellular composite materials
References [edit]
- ^ "Foam". Merriam-Webster. Archived from the original on 2014-12-09.
- ^ D. Weaire, South. Hutzler, "The Physics of Foams", Oxford Academy Press, 1999, ISBN 0198510977, ISBN 978-0-1985-1097-0
- ^ I. Cantat, South. Cohen-Addad, F. Elias, F. Graner, R. Höhler, O. Pitois, F. Rouyer, A. Saint-Jalmes, "Foams: structure and dynamics", Oxford Academy Printing, ed. S.J. Cox, 2013, ISBN 9780199662890
- ^ Lucassen, J. (1981). Lucassen-Reijnders, East. H. (ed.). Anionic Surfactants – Concrete Chemistry of Surfactant Action. NY, Usa: Marcel Dekker.
- ^ Morgan, F. "Existence of To the lowest degree-perimeter Partitions". arXiv:0711.4228.
- ^ Queheillalt, Douglas T.; Wadley, Haydn Northward.G. (Jan 2005). "Cellular metal lattices with hollow trusses". Acta Materialia. 53 (2): 303–313. Bibcode:2005AcMat..53..303Q. doi:10.1016/j.actamat.2004.09.024.
- ^ Kooistra, Gregory West.; Deshpande, Vikram S.; Wadley, Haydn N.G. (August 2004). "Compressive behavior of age hardenable tetrahedral lattice truss structures made from aluminium". Acta Materialia. 52 (14): 4229–4237. Bibcode:2004AcMat..52.4229K. doi:ten.1016/j.actamat.2004.05.039.
- ^ a b Bikerman, J.J. "Germination and Structure" in Foams New York, Springer-Verlag, 1973. ch 2. sec 24–25
- ^ "The Foam" (PDF). IHC News. January 2009. Archived from the original (PDF) on 2013-12-12. Retrieved 2013-12-09 .
- ^ Wilson, A.J, "Principles of Foam Formation and Stability." Foams: Physics, Chemistry, and Structure. New York, Springer-Verlag, 1989, ch 1
- ^ I. Roland, Thousand. Piel, 50. Delattre, B. Evrard International Journal of Pharmaceutics 263 (2003) 85–94
- ^ C. Lemarchand, P. Couvreur, Thou. Besnard, D. Costantini, R. Gref, Pharmaceutical Research, 20-eight (2003) 1284–1292
- ^ O. Mengual, M. Meunier, I. Cayre, K. Puech, P. Snabre, Colloids and Surfaces A: Physicochemical and Engineering Aspects 152 (1999) 111–123
- ^ P. Bru, 50. Brunel, H. Buron, I. Cayré, X. Ducarre, A. Fraux, O. Mengual, Yard. Meunier, A. de Sainte Marie and P. Snabre Particle sizing and characterisation Ed T. Provder and J. Texter (2004)
- ^ Wang, Shuo; Austin, Peter; Chakrabati-Bong, Sumana (2011). "Information technology's a maze: The pore structure of bread crumbs". Journal of Cereal Science. 54 (2): 203–210. doi:ten.1016/j.jcs.2011.05.004.
- ^ a b Gibson, Ashby (1999). Cellular Solids: Structure and Properties. Cambridge, U.k.: Cambridge University Press. ISBN9781316025420.
- ^ Wu, Qiong; Andersson, Richard L.; Holgate, Tim; Johansson, Eva; Gedde, Ulf W.; Olsson, Richard T.; Hedenqvist, Mikael South. (2014). "Highly porous flame-retardant and sustainable biofoams based on wheat gluten and in situ polymerized silica". Journal of Materials Chemical science A. 2 (48). 20996–21009. doi:10.1039/C4TA04787G.
- ^ Reyes-Labarta, J.A.; Marcilla, A. (2008). "Kinetic Study of the Decompositions Involved in the Thermal Degradation of Commercial Azodicarbonamide". Periodical of Practical Polymer Science. 107 (1): 339–346. doi:x.1002/app.26922. hdl:10045/24682.
- ^ Reyes-Labarta, J.A.; Marcilla, A. (2012). "Thermal Treatment and Degradation of Crosslinked Ethylene Vinyl Acetate-Polyethylene-Azodicarbonamide-ZnO Foams. Complete Kinetic Modelling and Analysis". Industrial & Engineering Chemistry Research. 51 (28): 9515–9530. doi:10.1021/ie3006935.
- ^ Reyes-Labarta, J.A.; Marcilla, A. (2008). "Differential Scanning Calorimetry Assay of the Thermal Handling of Ternary Mixtures of Ethylene Vinyl Acetate, Polyethylene and Azodicarbonamide". Periodical of Practical Polymer Science. 110 (5): 3217–3224. doi:10.1002/app.28802. hdl:10045/13312.
- ^ Reyes-Labarta, J.A.; Olaya, M.Thousand.; Marcilla, A. (2006). "DSC Study of the Transitions Involved in the Thermal Treatment of Foamable Mixtures of PE and EVA Copolymer with Azodicarbonamide". Journal of Applied Polymer Scientific discipline. 102 (iii): 2015–2025. doi:ten.1002/app.23969. hdl:10045/24680.
- ^ Ashida, Kaneyoshi (2006). Polyurethane and related foams: chemistry and technology. CRC Press. pp. 79–81. ISBN978-1-58716-159-nine. Archived from the original on 2017-02-17.
Literature [edit]
- Thomas Hipke, Günther Lange, René Poss: Taschenbuch für Aluminiumschäume. Aluminium-Verlag, Düsseldorf 2007, ISBN 978-3-87017-285-v.
- Hannelore Dittmar-Ilgen: Metalle lernen schwimmen. In: Dies.: Wie der Kork-Krümel ans Weinglas kommt. Hirzel, Stuttgart 2006, ISBN 978-3-7776-1440-iii, Due south. 74.
External links [edit]
| | Look upwardly cream in Wiktionary, the free dictionary. |
| | Wikimedia Commons has media related to Foam. |
- Andrew One thousand. Kraynik, Douglas A. Reinelt, Frank van Swol Structure of random monodisperse cream
- D. L. Weaire, Stefan Hutzler (1999) The Physics of Foams
- Moriarty, Philip (2010). "Foam Physics". 60 Symbols. Brady Haran for the University of Nottingham.
Source: https://en.wikipedia.org/wiki/Foam




























0 Response to "Is It Best to Use Variable Speed for Foam Houses"
Postar um comentário